Probiotic Compositions Containing Bacteriophage to Treat or Prevent Psoriasis
- 領域
- Therapeutic Biologics
- Patent
- IP00815
Key Problem and Market Opportunity
- Psoriasis is a chronic skin disease that affects 150 to 250 million people worldwide. In most developed countries prevalence is between 1.5 and 5%. Psoriasis has an unpredictable course of symptoms, a number of external triggers and significant comorbidities, including arthritis, cardiovascular diseases, metabolic syndrome, inflammatory bowel disease and depression.
- For psoriasis treatment, the global market size was valued at USD$11.3 billion in 2016. It is anticipated to post a CAGR of 9.4% for the next five years.
- Psoriatic skin infections are usually treated by antibiotics which can result in antibiotic resistance. New treatments include TNF or IL Inhibitors; however, are expensive and only control symptoms temporally (Figure 1).
Key Advantages of the Technology
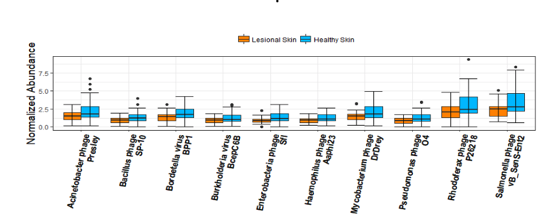
Skin microbiomes were investigated in patients with psoriasis and in healthy family controls. Top ten most abundant phage species among lesional skin and healthy skin were identified. These species were significantly more abundant in healthy skin than lesional skin, suggesting an association between phage species and psoriatic lesions ().
Moreover, further experimental data demonstrated that two phages species Acinetobacter phage Presley and Pseudomonas phage O4 have the capacity to suppress host bacteria linked to lesional skin.
This invention presents potential probiotic phage therapies for skin health in psoriasis which can restore the equilibrium of the skin microbiome in treated lesion areas.
Further Details
- Bacteriophage of the Skin Microbiome in Patients with Psoriasis and Healthy Family Controls31787-7/fulltext)
Benefits
- Suppresses unhealthy bacteria on psoriatic skin leading to healthier skin
- Reduces antibiotic use result in reduced antimicrobial resistance
- Cost-effective
Potential Product and Services
- Skin care product
- Topical therapies for psoriatic skin
Development Status and IP Strength
Patents
- PCT patent application No. PCT/CN2019/110309
IP Status
- Patent application submitted